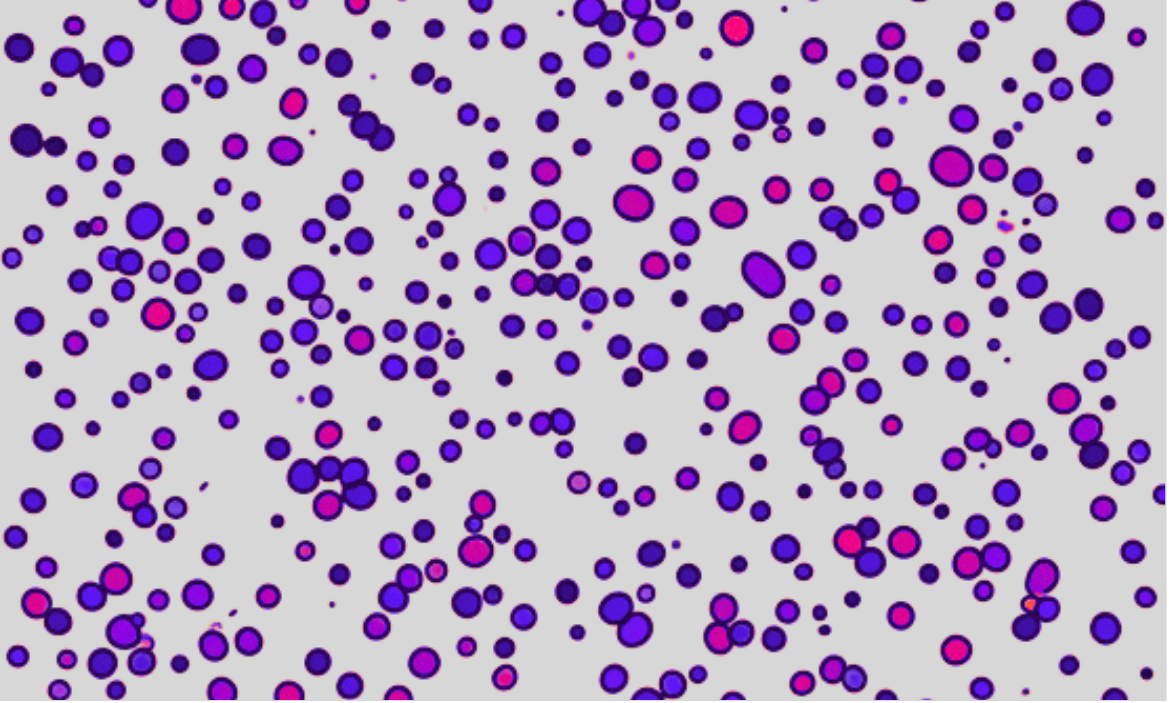
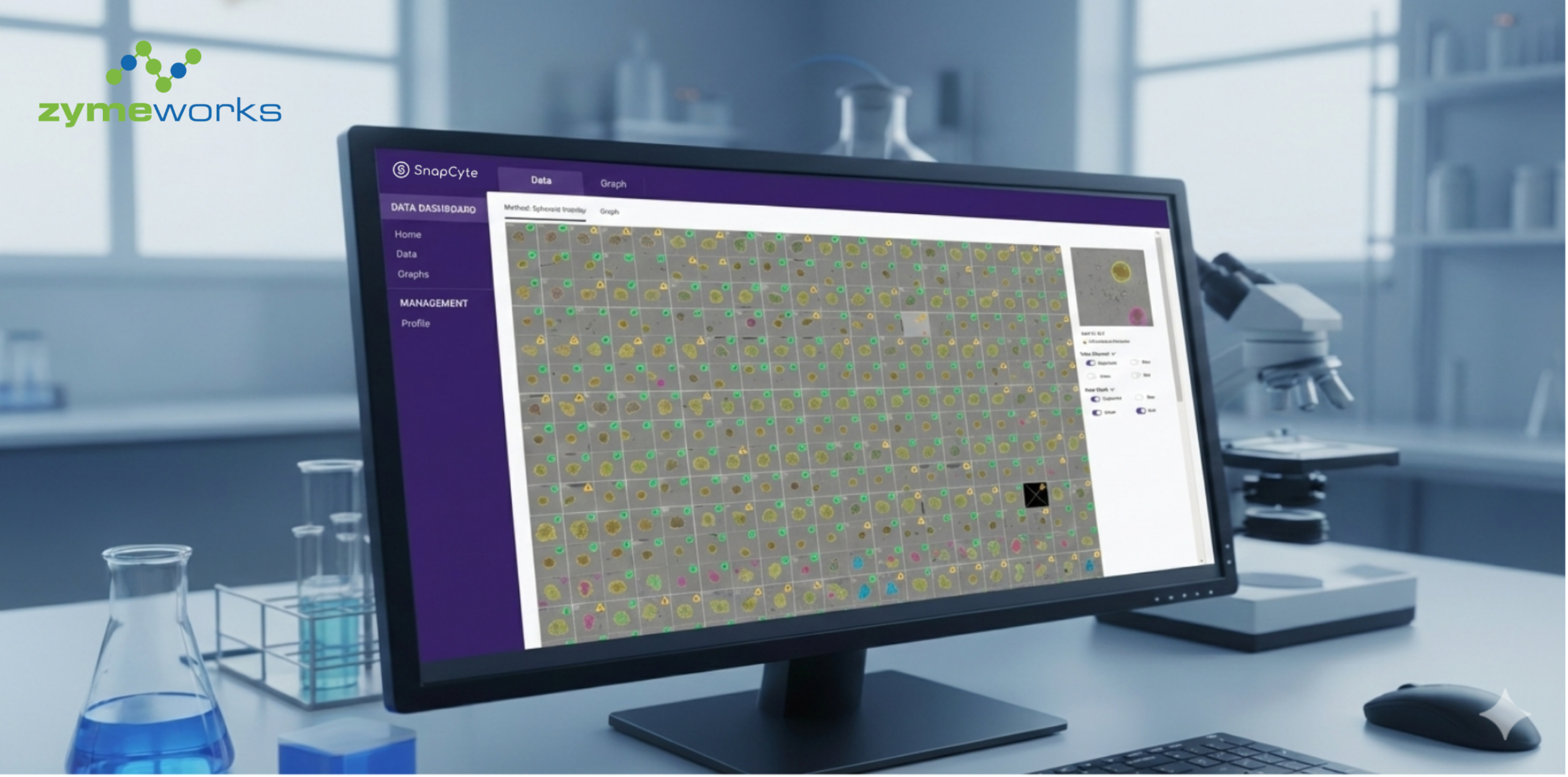
Summary
Cell confluency is a critical parameter in mammalian cell culture, and knowing the confluency level is important for controlling and optimizing cell growth, behaviour, and downstream applications. How is it defined, what affects it, and why is it so important to get just right? We explain all of that in this blog post. As datasets grow larger and more complex, consistent and objective analysis of cell confluency has become increasingly important.
Definition
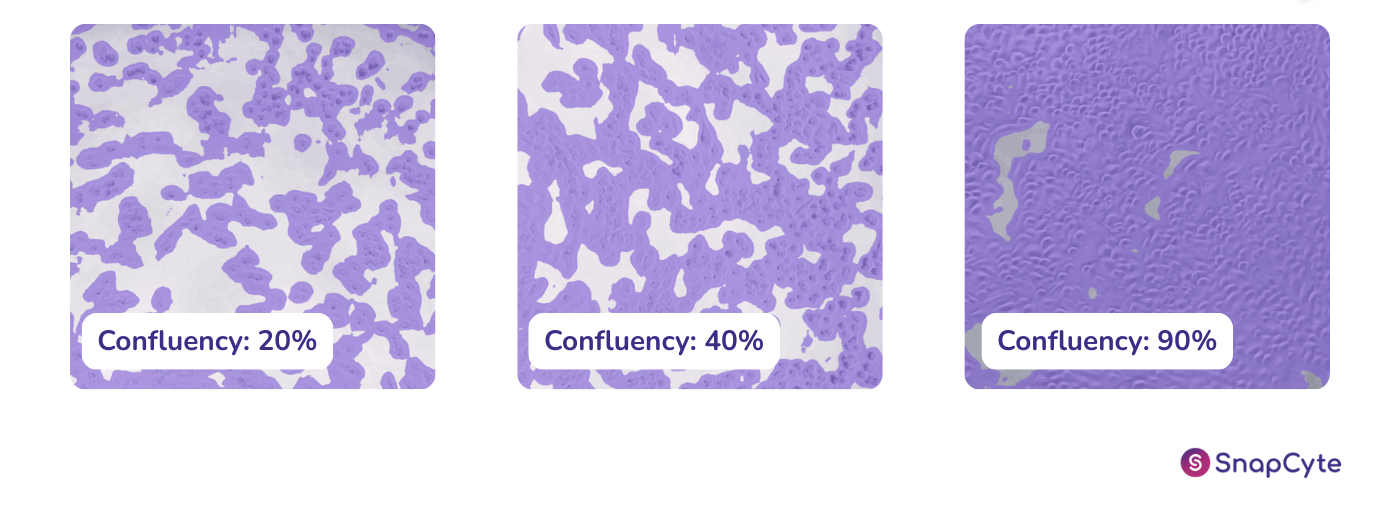
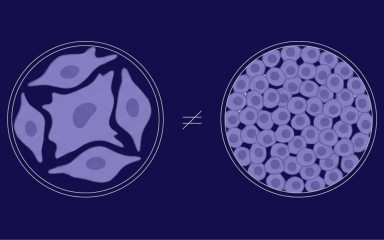
Cell confluency refers to the degree to which a culture of adherent cells in a dish or flask has filled a given area, typically measured as a percentage of the surface area covered by the cells. In other words, it’s the degree to which a culture dish or flask is “full” of cells. It is a very important parameter in cell culture and is used to determine when cells are ready to be subcultured, harvested, transfected, or otherwise used for experiments. The measurement of cell confluency is an essential step in maintaining healthy cell cultures and ensuring accurate experimental results.
Why Measuring Cell Confluency Accurately is Critical
Confluency is an important consideration in cell culture as it can affect the growth and behavior of cells in culture. High confluency can lead to issues such as reduced proliferation, increased cell death, and changes in cell behaviour. Cell confluency can be controlled by adjusting the seeding density of cells, or by removing cells via subculture. This can affect the outcome of experiments and the quality of the cells being used for research, therapy, or other applications.
The Six Main Reasons Why Cell Confluency is Important:
- Consistency: A confluent culture allows for a consistent and reproducible experiment, as all the cells are at the same stage of growth and division.
- Optimal growth conditions: Cells at confluency are typically grown under optimal conditions, which can minimize variability and improve the quality of the data generated from the experiment.
- Studying cell-cell interactions: Confluent cell cultures allow scientists to study the interactions between cells in a more natural and physiologically relevant manner, as the cells are in close proximity to one another and are able to interact through cell-cell junctions and signaling pathways.
- Synchronization: Confluent cell cultures can be used to synchronize cells for further experiments.
- Scalability: Confluent cell cultures can be used to scale up the production of cells for applications such as cell-based therapies and bio-manufacturing.
- Monitoring cell growth and proliferation: The percentage of confluency can be used as a measure of cell growth and proliferation, allowing scientists to monitor the growth of cells over time and make adjustments to the culture conditions as needed.
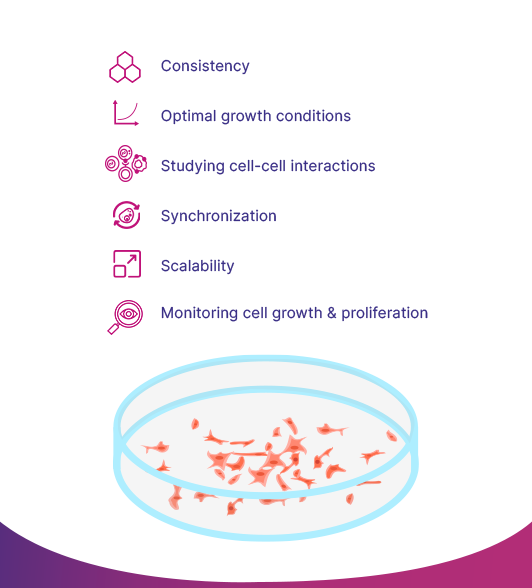
Regularly measuring the confluency is also important because when cells are grown in culture, they are often used for downstream applications such as protein production, gene expression analysis, and cell-based assays. The confluency level can affect the outcome of these downstream applications, so knowing the confluency level allows researchers to control and optimize the conditions for these applications.
Additionally, in many cell culture systems, cell confluency is used as a parameter to determine when to split or pass the cells. The cells are usually split when they reach a certain confluency level, so that they can continue to grow and divide without becoming too crowded and overgrown. Therefore, knowing the confluency level is essential for maintaining the health and vitality of the cells in culture.
Why Cell Confluency Measurement Becomes Subjective at Scale
In practice, cell confluency is rarely assessed from a single image or condition. Large experiments often include multiple replicates, timepoints, and treatment groups, making consistent interpretation difficult. Visual estimation and manual thresholds can introduce variability, especially when differences between conditions are subtle.
How to Measure Cell Confluency in Mammalian Cell Culture
There are several methods for measuring cell confluency, each with their own advantages and disadvantages. For a comparison of measuring methods, check out our new eBook: How to Measure Cell Confluency.
Manual/Visual Estimation (aka. 'Guesstimation')
One common method is manually estimating cell confluency using a microscope and visually inspecting the cell culture (sometimes referred to as ‘guesstimation’ due to the not so accurate data outcome). This method involves estimating the area of a cell culture flask or dish covered by cells and comparing it to the total surface area, resulting in a confluency percentage. The disadvantage of this method is that it is time-consuming and subject to observer bias. In many workflows, confluency estimation is closely tied to cell counting methods used to assess growth and viability.
Colorimetric Assays
A second method for measuring cell confluency is the use of cell-specific dyes or stains. These dyes or stains bind specifically to certain cell components, such as nuclei, and can be used to visualize the cells in a culture. By analyzing the intensity of the dye or stain, it is possible to estimate the number of cells in a culture. Although this method is more accurate than visual estimation or image analysis, it is also more expensive and time-consuming.
Image-Based Analysis Software
A number of tools offer image-based confluency measurements. These either consist of a software package that analyzes images taken from a microscope, or are integrated instruments that image and analyze your cell culture directly.
Digital image analysis software can be used to automatically quantify cell confluency from microscope images. These software programs can recognize cells and calculate the ratio of the cell-covered area to the total area, providing a more objective measurement than manual estimation. This method is more objective than manual counting, but it can be affected by variations in cell size, shape, and colour.
AI-Based Analysis Algorithms
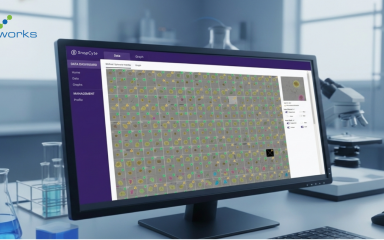
The most recent development in cell confluency tools is the incorporation of AI-based algorithms to analyze cell culture images. These tools apply the same analysis rules across all images, helping reduce variability caused by manual interpretation. They require no manual input and are a useful way to train students when initially wanting to learn to visually estimate a cell culture’s growth rate and overall health.
SnapCyte® is one example of an AI-based platform designed to support objective confluency analysis directly from microscopy images, without staining or specialized hardware.
What does 50% Confluency Mean?
50% confluency means that half of the surface area of the culture dish or flask is covered by cells. This can be determined by visually inspecting the culture under a microscope and estimating the percentage of the surface area that is covered by cells or by measuring the cell density. 50% confluency is often used as a benchmark for cell culture experiments as it is a point at which the cells are considered to be well-spread and actively growing, but not yet overcrowded. However, it’s important to note that the confluency percentage can vary depending on the type of cell and the culture conditions, so it’s important to establish what confluency means in your specific cell line and experiment.
How do you Know if a Cell Culture is Confluent?
A confluent cell culture refers to a condition in which the cells have reached a state of maximal density and have completely covered the surface of the culture dish. It measures as 100% confluency, and can be determined by visual inspection, where the cells appear to be tightly packed and there are no visible areas of the dish without cells. In addition to visual inspection, the confluency of a cell culture can also be quantitatively measured using an image-based system or by running a colorimetric assay.
In conclusion, measuring cell confluency is an essential step in cell culture and must be done with care and precision. There are several methods for measuring cell confluency, each with their own advantages and disadvantages. The choice of method will depend on the specific requirements of the experiment, including the type of cells, the available resources and the level of precision required. It’s important to note that cell confluency is not only a measure of cell number, but also a measure of cell health and activity, which can be impacted by various factors such as the quality of the cell culture and the timing of the harvest. Therefore, monitoring cell confluency is an important step for maintaining healthy cell cultures and ensuring accurate experimental results.
Manual vs AI-Based Confluency Measurement
Manual / Visual Estimation
- Subjective and user-dependent
- Limited reproducibility across users and timepoints
- Not scalable to large datasets or multiple experimental conditions
- Fast for quick culture health checks
AI-Based Confluency Analysis
- Applies the same analysis rules across all images and conditions
- Reduced user-dependent bias and variability
- Handles morphological and imaging variability
- Scalable to large, high-throughput datasets
Want to try AI-powered cell confluency analysis?
SnapCyte™ works with your existing microscope images.