Summary
Cell doubling time tells you how quickly a cell population grows under specific culture conditions. Along with passage number and population doubling level, it helps researchers track cell health, compare experiments, and identify culture drift before it affects results.
In this guide, we explain how to calculate cell doubling time, when the formula is valid, how passage number and PDL differ, and how image-based tools can make growth tracking more consistent.
What is doubling time?
In simple terms, doubling time definition is the period it takes for a population of cells to double in number. For example, if you seed 50,000 cells in a dish and after 24 hours you have 100,000 cells, then the doubling time under those specific conditions is 24 hours.
It reflects the growth rate of cells and it can tell you a lot about:
- Cell health, Healthy, actively proliferating cells have doubling times typical for their type. Deviations may suggest stress, senescence, or contamination.
- Experimental conditions: Media composition, oxygen levels, or the presence of drugs can speed up or slow down doubling time.
- Cell type differences: A rapidly dividing cancer cell line might double every 18–24 hours, while primary fibroblasts may take several days.
- HeLa → ~20–24 h (PMID: 28705234)
- PC3 → ~30–40 h (PMID: 33910833)
- LNCaP → ~60–72 h (PMID: 6831420)
- Primary fibroblasts → ~48–72 h (PMID: 36980290)
When is doubling time valid?
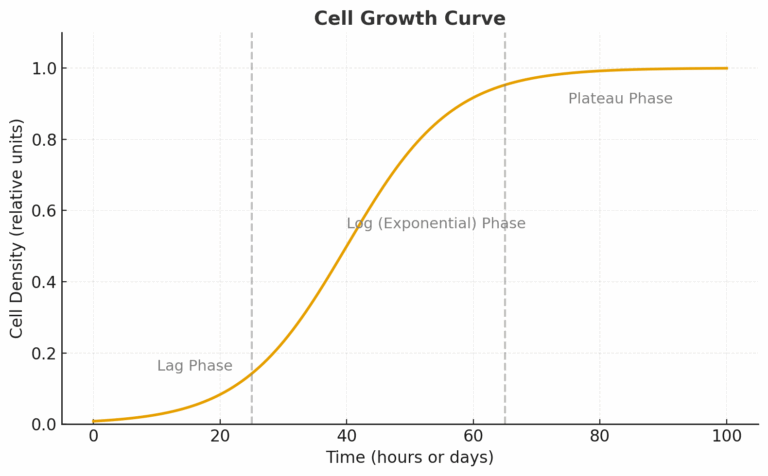
You should only measure doubling time during the exponential phase, it’s the only point where growth is truly proportional. In addition, doubling time is an average, some cells divide faster or slower than others, and shifts in subpopulations over passages affect the result.
The Growth Curve Phases:
Phase | Description |
Lag Phase | Cells adapt to new conditions; little division. |
Log (Exponential) Phase | Cells divide at a constant rate, this is when DT is valid. |
Plateau Phase | Growth slows as nutrients and space run out. |
Doubling time calculator
Use this formula to calculate cell doubling time based on growth between two points.
Doubling time (Td) = (t₂ − t₁) × ln(2) / ln(N₂ / N₁)
where:
N₁ = initial cell number (or confluency %)
N₂ = final cell number (or confluency %)
(t₂ − t₁) = elapsed time
Example:
If your cells grow from 1×10⁵ to 8×10⁵ in 48 hours,
Td = 48 × ln(2) / ln(8×10⁵ / 1×10⁵) = 16 hours.
You can use the calculator below to obtain the same result automatically by entering your cell count values and time intervals.
Doubling Time Calculator
Td = t × ln(2) / ln(Nt / N0)
What this does
How to collect data for doubling time?
- Direct Cell Counts
Hemocytometer, automated counter, or image-based adherent counts (e.g., SnapCyte Cell Count). - Confluency Measurements
Estimate % area coverage using live-cell imaging , fast and non-destructive. (e.g. SnapCyte Confluency) - Colorimetric Assays
MTT, XTT, or WST-1 to measure metabolic activity (less precise but scalable).
What does a change in doubling time mean?
Doubling time can serve as an early warning for cell culture problems:
- If a cell line doubling time changes by more than 20–30% from baseline, that’s a red flag.
- Causes include senescence, genomic instability, contamination, or adaptation to artificial conditions.
- Rule of thumb: When doubling time shifts significantly, return to earlier passage numbers or thaw a new stock.
- Because growth rate is tied to genetic stability, doubling time acts as a low-cost proxy for culture integrity
Routine measuring of doubling time:
Traditionally, measuring doubling time required destructive assays, but modern imaging tools make it routine.
For example, with SnapCyte:
- Take a picture when you plate cells.
- The software calculates cells per cm² from adherent counts or confluency %.
- Capture images at intervals.
- Use cell growth assay to measure adherent cell count and confluency to calculate doubling time in the calculator above.
This approach avoids detaching or sacrificing cells, critical for rare or primary cultures where material is precious.
What to do when doubling time changes?
A significant deviation (>20%) from your baseline doubling time is a red flag:
- Check media quality and serum lot
- Verify seeding density and CO₂ calibration
- Inspect morphology and contamination
If the issue persists, discard the culture and revert to an earlier passage.
Applications beyond cell culture
- Oncology: Doubling time correlates with tumor aggressiveness; changes in vitro may reflect altered disease biology.
- Biomanufacturing: Shorter doubling times directly increase yields in vaccine, viral vector, and protein production.
- Stem Cell Therapy: Regulatory agencies require strict passage and doubling time tracking to ensure safety and potency.
What is Passage Number?
Every time you transfer cells into a new flask with fresh medium, you create a new passage. The passage number of a cell line records how many times this has happened since the culture was first established or thawed.
Why passage number matters?
Over time, cells change, genetically, epigenetically, and phenotypically. Tracking passage number helps you monitor these changes and keep experiments consistent.
At high passages, cells may:
- Genetic and phenotypic drift: The cell population can accumulate genetic and epigenetic changes over time. This can alter cell morphology, gene expression, and overall behavior, causing the culture to no longer accurately represent the original cell line.
- Experience selection pressure: In a heterogeneous cell population, faster-growing cells may out-compete and eventually dominate the culture, fundamentally changing the population’s characteristics.
- Altered function: The ability of cells to function as intended can change. For example, some cell lines lose their differentiation capacity or change their drug responsiveness after extended passaging.
- Reduced health: High-passage cells may show altered growth rates, decreased viability, or signs of senescence, especially in primary cell cultures that have a finite lifespan.
⚠️ Using high-passage cells without documentation is one of the most common causes of irreproducible results.
How to track cell line passage number?
- Establish a Master Cell Bank: Freeze multiple vials of low-passage, authenticated cells (e.g., ATCC, DSMZ).
- Define a Passage Limit: Decide in advance the maximum passage for each line (e.g., ≤20 for immortalized lines, ≤7 for primary cells).
- Document Consistently: Record thawing date, media, split ratio, morphology, and any changes in behavior.
- Know What Counts as a New Passage: A new passage is counted only after cells are subcultured, not after thawing a new vial.
- Standardize How You Passage Your Banked Cells: Always passage your banked cells using consistent criteria and timing. For example: Passage them when they reach ~80% confluency , and don’t guess it! Measure it (it’s 2025, use your imaging tools 😊). Seed cells consistently at the recommended confluency or cell density for your specific line (e.g., 30%). Maintaining a predefined and uniform confluency at both seeding and harvesting ensures reproducible growth behavior across passages.
💡 Tips:
- Always include passage number in your methods section, reviewers notice.
- Maintaining a predefined and uniform confluency at both seeding and harvesting makes Population Doubling Level (PDL) calculation and tracking straightforward , we’ll explore how to do that later.
Want a faster way to measure doubling time?
Try SnapCyte®, our AI-powered tool for measuring confluency, adherent cell count, and cell growth trends from microscopy images.
What is Population Doubling Level (PDL) ?
While passage number tells you how many times you handled the cells, and doubling time shows how fast they divide, neither reveals how many total doublings have actually occurred. That’s where Population Doubling Level (PDL) comes in.
What is the definition of population doubling time?
PDL represents the cumulative number of population doublings since the culture’s establishment, the true biological age of your cells.
Formula:
PDL=3.32×log10(N2/N1)
where:
N1 = initial cell number
N2=final cell number
This formula calculates how many times the cell population has doubled between two passages. To find the cumulative population doubling level across several passages, add each interval’s value:
Cumulative PDL=∑3.32×log10(N2/N1)
Why PDL matters more than passage number?
Concept | Passage Number | Population Doubling Level |
What it measures | Subculture count | Actual doublings |
Sensitive to split ratio | Yes | No |
Biological meaning | Rough handling record | True replicative age |
Used for | Routine tracking | Primary cells, senescence, quality control |
Example:
- A 1:2 split = 1 doubling
- A 1:10 split = 3.3 doublings.
Both increase passage by one, but the PDL increase is very different. PDL corrects this bias, providing a standardized measure of cellular age across labs and experiments.
How to consistently track PDL?
To make PDL meaningful:
- Seed and harvest at the same confluency each passage (e.g., 20–30% → 90–100%).
- Use consistent vessel sizes and surface areas.
- Record seeding density, harvest count, and passage interval.
Once growth stabilizes, each passage adds a roughly constant ΔPDL (e.g., +3.0 per passage). Then you can estimate:
Cumulative PDL=Starting PDL+(Number of passages)×Average ΔPDL
PDL in Cell Banking
Recording the PDL at the time of freezing ensures:
- Traceability between vials
- Consistency between users or labs
- Genetic and phenotypic stability over time
Regulated environments (GMP, stem cell therapy, etc.) require both passage number and PDL for every banked cell lot.
Further Reading
Passage number is a major contributor to genomic structural variation in mouse embryonic stem cells
Liu P, Smith CL, Huang S, Maher C, Ganesan S, et al.
Genome Res. 2014; 24(9):1481–1490.A beginner’s guide to cell culture: practical advice for experimental design and culture maintenance
Weiskirchen S, Weiper K, Weiskirchen R.
Cells. 2023; 12(5):682.The Effect of Repeated Passaging on the Susceptibility of Human Proximal Tubular HK-2 Cells to Toxic Compounds
Handl J, Shabbir M, Danyal T, et al.
Cells. 2020; 9(10):2261..In Vitro Research Reproducibility: Keeping Up High Standards
Hirsch C, Schildknecht S.
Front Pharmacol. 2019; 10:1484.Cell Culture: Growing Cells as Model Systems In Vitro
Review / Methods chapter, PubMed Central (PMC7149418)

Nader Al Nakouzi, PhD
I am a cancer biologist and translational researcher with experience in cell culture, microscopy, image-based assays, and therapeutic development. My work focuses on understanding complex biological systems and improving how experimental data is measured, interpreted, and translated into meaningful decisions.